The symptoms started out as mild stomach issues for Appleton resident Bret Carroll.
But when Carroll, a healthy and active 45-year-old professional pilot, found a lump on his abdomen in 2011, it signaled an enlarged spleen.
“They went through a process of doing CAT scans and blood work,” Carroll said. “They started narrowing a diagnosis down, and they kept eliminating the more benign options.”
Then all options were eliminated but one: mantle cell lymphoma, a rare cancer of the immune system. Carroll reported immediately to his first chemotherapy treatment and was given a prognosis of about two years.
“There was a pamphlet they gave us and it specifically mentioned that mantle cell was incurable,” Carroll said. “So it struck even harder than a diagnosis of cancer.”
A father of three high school students, Carroll considered what he wouldn’t live to see.
“It seemed like there were a lot of things we were going to miss out on that we still wanted to do,” he said. “The thought of not being able to see them graduate, it was very tough.”
Because he was in otherwise good health, doctors advised Carroll to pursue the most aggressive treatments available, with the promise that others would become available over time.
The next few years brought the highs and lows of treatment – the hopes and letdown of chemotherapy, stem cell transplants, radiation therapy and a clinical trial of a new drug that ended up producing extremely negative side effects, including severe weight loss.
With each roadblock, Carroll held out hope for what doctors initially told him – that new treatment options were coming.
“With the ups and downs over the years, it was disappointing to have setbacks, but it wasn’t like the initial shock of being diagnosed,” he said. “Even though the treatments themselves were a hardship, there was something to keep me going.”
The rise of CAR T-cell therapy

Meanwhile, a promising new approach to cancer treatment was emerging.
In recent years, immunotherapy – treatment that uses a patient’s own immune system to help fight cancer – has gained traction in the cancer treatment world.
It represents a departure from what have become common cancer treatments, such as transplants, chemotherapy and radiation therapy – methods that, while effective in killing cancer cells, also often damage healthy cells.
One particular immunotherapy approach, called CAR (chimeric antigen receptors) T-cell therapy, has emerged as a breakthrough technology over the past five years. CAR T-cell therapy genetically reprograms a person’s immune system to target cancer cells in a personalized way. The research community has increasingly begun recognizing it as an option for patients for whom other lines of treatment have failed – patients like Carroll.
By the fall of 2017, the previous clinical trial in which Carroll had participated initially proved effective against the cancer, but the dosage brought on severe side effects.
That’s when he learned of CAR-T therapy.
His prior experience gave him pause about trying another clinical trial, but Carroll recognized something different about this option. While other treatments offered a possible chance at remission and extending Carroll’s life, the CAR-T option held the promise of complete remission – a chance at a cancer-free life.
Carroll was told it carried the same amount of risk as some aggressive chemotherapy treatment options, with about 30 percent of CAR-T patients experiencing no symptoms at all and about 10 percent ending up in the ICU.
Encouraged by the results of clinical trials for other types of cancer, physicians saw it as the best option for Carroll.
“In the end, I decided to go with the CAR-T and hope for the best,” Carroll said.
In-house advantage
On the third floor of Froedtert Hospital, a lab houses the CliniMACS Prodigy, a device designed to manufacture cellular products used in cell-based therapies.
In 2014, Froedtert & the Medical College of Wisconsin was among the first U.S. medical centers to acquire the technology, purchased with philanthropic support from the MACC Fund and Children’s Hospital of Wisconsin Foundation. The focus at the time was on pediatric bone marrow treatment, but the machine proved to be useful in the emerging field of immunotherapy.

Medical College of Wisconsin
“We were seeing where the field was going and knowing that we need the infrastructure and resources in this lab to stay ahead in the field of immunotherapy,” said David Margolis, blood and marrow transplant program director for Children’s Hospital of Wisconsin and professor of pediatrics in the hematology and oncology division at MCW.
In the case of immunotherapy, the contained, cell-filtering desktop system collects a patient’s T cells and augments them with cancer-fighting genes, and the new cells are then infused back into the patient’s bloodstream.
Having the technology in-house has placed MCW at an advantage in the CAR-T therapy arena. Traditionally, medical centers have had to ship cells to large manufacturing plants, where they undergo genetic modifications and are sent back to be returned to the patient.
At the Froedtert campus, the whole process is completed on-site, cutting costs and production time in half.
“What we’re doing here is a point-of-care model, meaning instead of doing third-party shipping, we collect the cells here, use cutting-edge technology, the (CliniMACS) Prodigy device, to do the CAR T-cell production and genetic modification right here in our lab,” said Nirav Shah, principal investigator of the trial and an assistant professor of medicine in the hematology and oncology division at MCW.
Once collected, the reprogrammed cells can be infused back into the patient within 14 days.
Dual-targeted therapy
The timing of Carroll’s decision to pursue CAR-T therapy proved critical.

Lauren Anderson
In the fall of 2017, his cells were collected before complications from the previous drug and the return of his cancer sent him to the hospital.
“Things started to go downhill to the point where I was in the hospital with them trying to keep me alive long enough to get the processed T cells back,” Carroll said.
Carroll received his CAR T cell dose on
Oct. 31.
Carroll was administered a unique product developed by MCW researchers: a dual-targeted CAR T-cell therapy that targets two proteins on the cancer cells, called CD19 and CD20, both of which are prevalent in patients with mantle cell lymphoma. FDA-approved products only target one protein.
Almost immediately after receiving the cells, Carroll sensed improvements in his health.
“It was like any time you’re sick with some sort of infection, that’s how my body was reacting – some mild fevers, I was worn out, a little achy, but over a few days, I was feeling better and ready to get out of the hospital,” Carroll said.
Within a few days, evidence suggested the T cells had been effective.
Six weeks after receiving the CAR T cell dose, Carroll’s cancer was no longer detectable.
In December, a second participant in the clinical trial received a dose of CAR T cells, and a third patient began treatment earlier this year.
‘Survivorship is the goal’
The cancer treatment journey has a way of tempering expectations. For Carroll, it isn’t the first time cancer has disappeared from view, but he’s optimistic.
“I have been here before a few times,” he said. “This time does feel different, but to a great extent I have the attitude of ‘we’ll see.’ The longer it lasts, the more confident I’ll become that it’s really for good this time.”

Week to week, Carroll has experienced an increase in his energy, though he still deals with the side effects of the previous clinical trial.
Carroll is considered to be in clinical remission, recovering from years of therapy, Shah said.
With the pace of medical appointments expected to decelerate to annual check-ins, Carroll said he is ready to return to his pre-cancer routines of flying, exercising and spending time with family.
No longer tied to the hospital, Carroll and his wife, Theresa, might even be able to take a vacation, he said.
“Now, we don’t have to think about cancer that much,” he said. “We can focus on the side effects and hopefully we can get over those and I can be back to a full and healthy life.”
The cancer team is also encouraged by his results. Shah called them “phenomenal.”
Still, the word “cure” is spoken with caution.
“Survivorship is the goal,” Margolis said. “I learned a long time ago from the kids and parents we take care of that the word ‘cure’ has a certain aura around it. Our patients always live with the dread of cancer coming back, of side effects, so we aim for 100 percent quality of life survival.”
Investment in research
Clinical trial data show cancer is a major research focus for MCW.
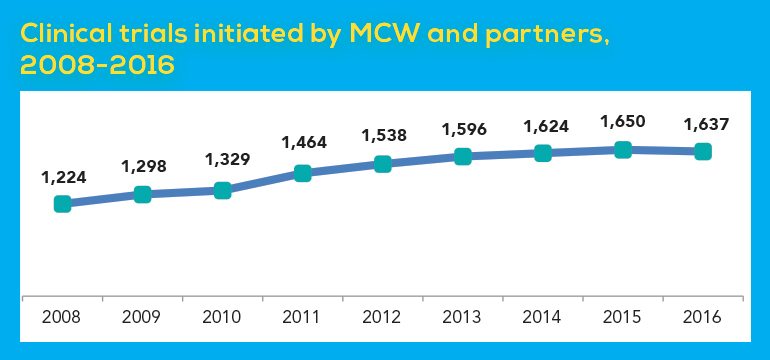
Faculty and staff have had a steadily increasing number of new clinical trials over the past decade – a jump of about 34 percent between 2008 and 2016, according to a recent Wisconsin Policy Forum report. In 2016, there were 1,637 clinical trials initiated by MCW and its partners.
Over the past decade, 559 hematology oncology clinical trials have been approved by the MCW/Froedtert institutional review board, the most of any discipline. Together with surgical oncology and radiation oncology, they accounted for a combined total of 878 clinical trials over the past 10 years.
Still, the Wisconsin Policy Forum identifies medical research as a critical funding challenge for the institution moving forward, particularly as federal funding tightens.
MCW invests about $65 million to $70 million in research each year from internal funding sources, largely leaning on its clinical operations, and brings in about $160 million to $170 million of external funding annually.
“Many times, people think you make a profit off of your research enterprise and that’s simply not the case,” said Dr. John Raymond, president and chief executive officer of MCW.
While MCW’s research and development expenditures make it the largest research institution in the metro Milwaukee area, its R&D spending has declined each year since 2011. That bucks the national trend, in which university R&D expenditures inched up each year between 2011 and 2015.
Driving that decline has been the decrease in federal research funding flowing to MCW, which dropped by about 19 percent between 2011 and 2016. Federal funding to MCW peaked in 2011 at $134 million, and declined over the next three years before flattening out, explained at least in part by the expiration of a temporary infusion of federal funding through the American Recovery and Reinvestment Act between 2010 and 2013.
That places more pressure on MCW’s clinical operations to help maintain its roughly $230 million annual R&D investment, at a time when the Froedtert & MCW health network competes with non-teaching hospitals within larger consolidated health systems.
“It is a challenge for us,” Raymond said. “To have something vitally important like the research enterprise, one of our core missions, it requires a chronic, substantial investment each and every year.”
MCW has encouraged more collaboration among faculty to increase their competitiveness for securing external funding, and has narrowed in on several R&D priority areas, he said.
Maintaining a robust research arm, Raymond said, is key to retaining attraction and retention of high-quality faculty.
“That research enterprise is why we have such a large clinical enterprise,” Raymond said. “The clinicians come where they can work with scientists and where there are scientists who are interested in helping them render better health care.”
Future of immunotherapy
Still in the earliest clinical trial phase, the CAR T-cell research at MCW is currently aimed at defining the safety of the product and its effectiveness. Once those are determined, the next steps will be to scale up the research by conducting studies at other medical centers.
A separate pediatric clinical trial for CAR T-cell treatment at Children’s Hospital of Wisconsin is also in the works, expected later this year.

Lauren Anderson
“We’re very excited about Bret, but one patient doesn’t necessarily mean it will be a 100 percent response rate for other patients,” Shah said.
“There’s no doubt that people who have developed cancer in the last decade, some are alive because of these technologies, compared to the decade before,” he added. “The ultimate goal is to have every situation be like Bret’s – that amazing response that we hope is durable.”
The next frontier will be to explore its effectiveness in treating “solid” cancers, including breast and colon cancer.
“The big advancement will be if this moves outside of blood cancer and into other types of cancer,” said Bryon Johnson, professor of pediatrics in the hematology and oncology division at MCW and a member of the Cancer Center. “I think it will be forthcoming at some point in time, but the research has to continue.”
“Cancer immunotherapy, as we move forward, is going to involve a variety of different strategies,” Johnson said. “It’s probably not going to be one strategy that is the panacea. It is going to be a mixture of different types of Breast Cancer Treatment Services. This is exciting that this is one treatment of many.”
For example, this month, the Froedtert & MCW Cancer Network added another type of CAR-T therapy option, called Kite Yescarta, made available to patients with large B-cell lymphomas for whom two or more chemotherapy drugs haven’t been effective.
Margolis said the rapidly evolving field gives reason for optimism.
“The iceberg is immunotherapy, and CAR T cells are the tip of that iceberg that we are just starting to understand,” Margolis said. “What started out as Ford Model T became a Corvette. It’s a different brand. The good news for patients throughout the world is there will be different brands of CAR T cells for different cancers. We’ll continue to make improvements, just like the car industry does. And one of these days, we’ll have a Tesla.”

